The Mass of Atoms
DVD or VHS Format
 With the appearance of the
subject of atoms quite early in the K-12 curriculum, students are entitled to understand how we know the mass of a single atom or its equivalent,
Avogadro's number. However, in many widely-used textbooks, this important scaling factor between the daily world and the atomic world is either omitted
or is stated without any indication of how it is known.
With the appearance of the
subject of atoms quite early in the K-12 curriculum, students are entitled to understand how we know the mass of a single atom or its equivalent,
Avogadro's number. However, in many widely-used textbooks, this important scaling factor between the daily world and the atomic world is either omitted
or is stated without any indication of how it is known.
In the now classic experiment shown in The Mass of Atoms, Raymond Hertz and Charles Brewer of Monsanto's Mound Laboratory, find the mass of a single atom by using the simple relationship:
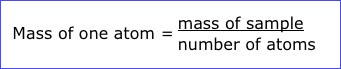
The film shows in detail how measurements to determine the mass of a sample and the number of atoms in the sample are carried out.
Produced in 1965 by Education Development Center of Newton, MA, for students of Introductory Physical Science (IPS), The Mass of Atoms is self-contained and can be profitably shown in a wide variety of courses, including high school chemistry and physics classes.
During The Mass of Atoms, a sample of radioactive polonium is massed and sealed in an evacuated quartz tube. It is then left to decay. The collection of alpha particles from the decaying polonium provides a sample of helium. After a three-week period, the mass of the helium sample accumulated is found and the rate of decay of the polonium is measured. From these data, the atomic masses of helium and polonium are determined. Throughout the film, the various laboratory techniques and precautions necessary for these measurements are shown.
This combination of The Mass of Atoms, Parts I and II is presented with the permission of Education Development Center, Inc., Newton, Massachusetts ©1965 Education Development Center, Inc. All rights reserved.
Re-mastered, 2004; Original, 1965
Running Time: 45 minutes 35 seconds
Price and Ordering Information
You may email us with specific questions or use our information request form on this website.
Available: Now

The new 9th Edition of Introductory Physical Science (IPS) continues the IPS tradition of guiding students to knowledge of physical science and the way scientific knowledge is acquired.
Inspect the table of contents and more details on our new edition of IPS.
